Summary:
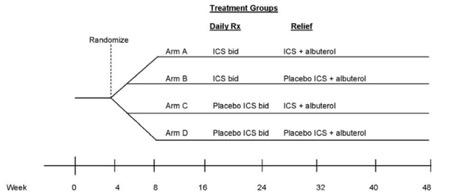
In children with mild persistent asthma who are under good control with low dose inhaled corticosteroid (ICS) (100 μg fluticasone b.i.d. or its equivalent) for at least 8 weeks, and are thus eligible for weaning from ICS therapy according to NAEPP guidelines, treatments for 44 weeks with:
- the same dose of ICS twice daily and use of ICS + albuterol as reliever;
- the same dose of ICS twice daily and use of placebo ICS + albuterol as reliever;
- placebo ICS twice daily and use of ICS + albuterol as reliever;
- placebo ICS twice daily and use of placebo ICS + albuterol as reliever do not differ in their effects on the time to first asthma exacerbation
Design:
TREXA is a 44-week randomized, double-blind, double-masked, four-treatment, parallel trial that will evaluate the weaning strategy that provides the best protection against the development of exacerbations in children whose asthma is acceptably controlled on a low dose of ICS (per NAEPP guidelines). Following the 4 weeks of the run-in period on a 1x dose of ICS (100 μg fluticasone b.i.d. or its equivalent), children who do not meet the definition of acceptable asthma control will be randomized to the parallel BADGER protocol; those who meet the definition of acceptable asthma control will be enrolled into the 44-week treatment phase of the study. The primary outcome measure will be time to first exacerbation requiring a prednisone course.
Population:
Childhood Asthma Research and Education (CARE) Network
Link:
Enrollment for TREXA began in November 2006 with a target sample size of 280 randomized children into the four treatment groups. The final patient visits occurred in February 2010 with a total sample size of 288 randomized children: combined (n=71); daily beclomethasone (n=72); rescue beclomethasone (n=71); placebo (n=74). Compared with the placebo group (49%, 95% CI 37–61), the frequency of exacerbations was lower in the daily (28%, 18–40, p=0.03), combined (31%, 21–43, p=0.07), and rescue (35%, 24–47, p=0.07) groups. Frequency of treatment failure was 23% (95% CI 14–43) in the placebo group, compared with 5.6% (1.6–14) in the combined (p=0.01), 2.8% (0–10) in the daily (p=0.009), and 8.5% (2–15) in the rescue (p=0.02) groups. Compared with the placebo group, linear growth was 1.1 cm (SD 0.3) less in the combined and daily arms (p<0.0001), but not the rescue group (p=0.26).The major publication appeared in The Lancet:
Martinez FD, Chinchilli VM, Morgan WJ, Boehmer SJ, Lemanske Jr RF, Mauger DT, Strunk RC, Szefler SJ, Zeiger RS, Bacharier LB, Bade E, Covar RA, Friedman NJ, Guilbert TW, Heidarian-Raissy H, Kelly HW, Malka-Rais J, Mellon MH, Sorkness CA, Taussig L. Preventing exacerbations in mild persistent childhoodasthma: Use of beclomethasone diproprionate as rescue therapy in a randomized, placebo controlled trial. The Lancet 2011; 377:650-657. PMID: 21324520

